 
What was the evidence that the cathode rays were particles with charge and mass? Remember to submit your answer to A 1 to your teacher as part of your Module 7: Lesson 1 Assignment.Ī 1. Read “Cathode-ray Experiments” on pages 754 to 755 of your physics textbook. 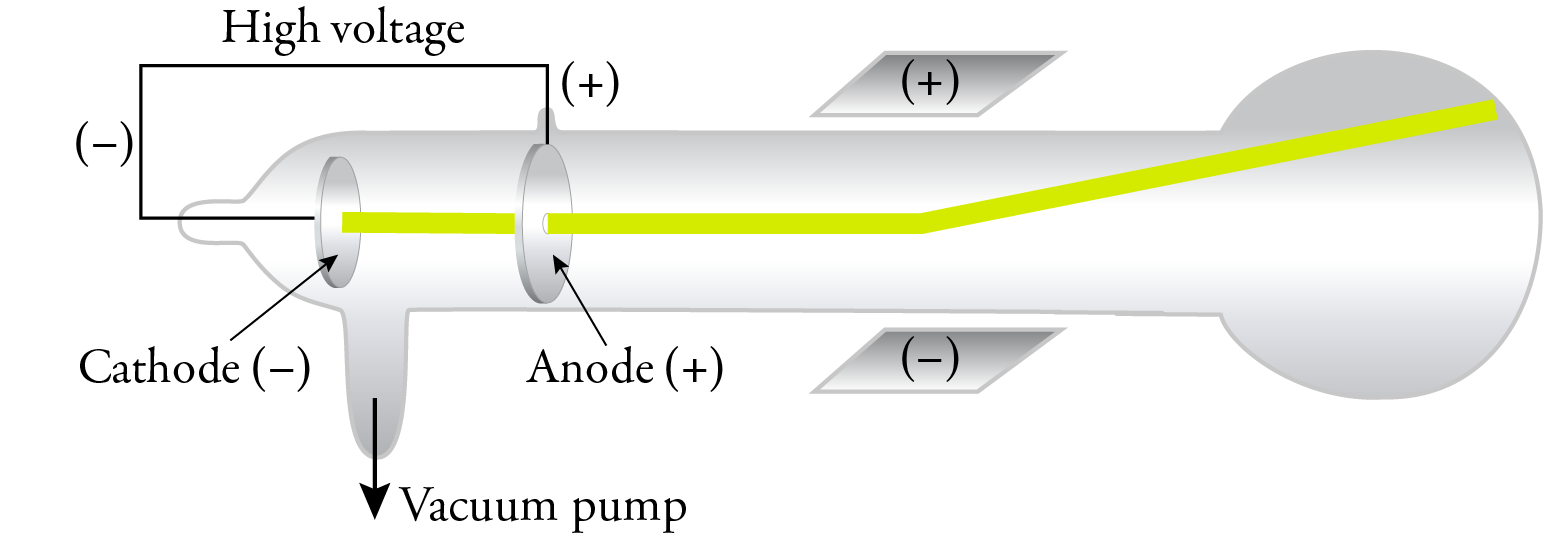
are deflected by magnetic and electric fields and have a negative charge.travel in straight lines and cause shadows.travel from the negative electrode to the positive electrode.The direction of deflection of the cathode rays further proved that they were, in fact, negative.īased on these observations, cathode rays Using the left-hand rule for moving electric charges in a magnetic field, he proved that the particles were negatively charged and were emitted by the cathode.Īrthur Schuster used a set of external charged plates to demonstrate that the cathode rays were affected by electric fields. This proved that cathode rays were composed of moving particles that had mass and momentum and were capable of doing work.Ĭrookes also demonstrated that cathode rays are deflected by a magnetic field. When the rays hit the paddles, they caused the wheel to move along a track inside the tube. Crookes placed a paddle wheel, which was free to rotate, in the path of the cathode rays. For this reason, gas discharge tubes are sometimes called Crookes tubes or, more commonly, cathode ray tubes (CRT). William Crookes proved that the emissions travelled from the cathode to the anode. Immediately, the cathode ray became the subject of intense experimentation. This diagram is an illustration of a simple gas discharge tube. 
Cathode ray: a free electron emitted by a negative electrode in a low-pressure environment 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
